Heat stress in pigs is defined as a physiological response to high environmental temperatures, where an animal is out of its thermoneutral zone and can no longer effectively regulate its body temperature. As a result, animal health, well-being, and performance are negatively affected.
The effect of heat stress in pigs
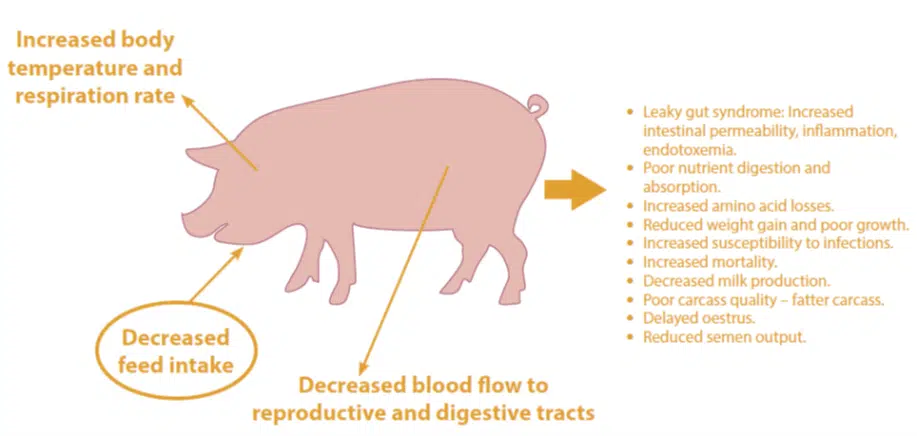
When pigs are exposed to high temperatures, several behavioural, physiological, and metabolic mechanisms are activated to increase heat dissipation to maintain the body temperature within normal physiological ranges (Figure 1). The most effective mechanism to reduce heat production is to lower the feed intake, and this drop in feed intake is accentuated as bodyweight increases.
It has been reported that pigs with a bodyweight of between 60 kg and 100 kg raised in south-eastern Brazil during summer, exhibit approximately 15% lower growth rates compared to pigs raised during winter. This effect can be explained in part by the decreased feed intake during the hot season.
Pigs have few, if any, functional sweat glands, and evaporation relies only on drooling and an increased respiration rate (panting). Consequently, they depend on radiation to dissipate heat. Young pigs are usually not affected by heat stress as much as larger pigs, because they are smaller and have a higher zone of thermal comfort. Piglets experience heat stress indirectly through a reduction in sow milk production (Black et al., 1993).
Increased energy and micronutrient utilisation, coupled with reduced feed intake during heat stress, can induce micronutrient deficiencies and altered metabolism, culminating in poorer growth performance.
Managing heat stress with zinc
Zinc is commonly supplemented in animal feeds to maintain health status and achieve optimal growth performance. Inorganic zinc, mainly zinc oxide (ZnO) and zinc sulfate (ZnSO4) is widely used for this purpose.
In practice, pharmacological levels of zinc have been widely used to improve the growth performance of young pigs (Carlson et al., 1999). Zinc has also been explored as a supplement for protecting animals against the adverse effects of heat stress (Sahin & Kucuk, 2003; Sahin et al., 2009).
The protective effects of zinc have been partially attributed to its antioxidant properties (Powell, 2000), while its growth-promotion effects are likely due to its functions as the co-factor of insulin, transcriptional factors, and metabolic enzymes (Lynch et al., 2001; Hedemann et al., 2006; Maret, 2013).
Studies conducted in humans and mice have shown that zinc is essential for normal intestinal-barrier function and the regeneration of damaged gut epithelium (Alam et al., 1994). Research has also shown that zinc acts primarily at the tight junction level to decrease intestinal permeability, while maintaining intestinal integrity.
A follow-up study was conducted on 59 gilts to evaluate the effect of feeding varying levels of zinc for 28 days prior to exposure to both acute (24-hour periods) and chronic heat stress for seven days. Results showed that the maximal benefit in transepithelial resistance – the permeability of the gut – was found in gilts fed complexed zinc at 220 ppm.
According to Baumgard (2016), this research shows that feeding zinc improves intestinal integrity, that gut integrity during heat stress can be improved with zinc levels at 120 ppm, and that the source of zinc is important.
The role of chromium
The involvement of insulin in the heat stress response was suspected following observations of increased mortality of diabetic patients during heat waves (Schuman 1972; Semenza et al., 1999). The specific mechanism whereby insulin counteracts the effects of heat stress is not fully known, but it is supposedly partly due to improved regional blood flow, which augments radiant heat loss.
Therefore, compounds that improve insulin sensitivity may be beneficial during heat stress. Chromium is an essential mineral and has been conditionally included in pig diets to improve growth performance and carcass traits. Chromium augments the receptor-binding of insulin, augmenting insulin sensitivity via the chromium-containing protein, chromodulin (Davis et al., 1997).
Reduced plasma cortisol is a typical metabolic response to chromium supplementation in livestock, especially when animals are under stress (Chang & Mowat, 1992; Samanta et al., 2008; Zha et al., 2009; Hung et al., 2014). Chromiumregulated insulin action may occur via altered cortisol concentrations (Borgs & Mallard, 1998) because cortisol secretion increases when the animal is under stress and acts as an insulin antagonist.
In this context, cortisol increases plasma glucose concentrations and slows down glucose utilisation by peripheral tissues, which indicates induced insulin resistance. The maintenance of plasma glucose homeostasis is a complex process involving many hormones and genes. A proper response of the insulin-signalling pathway genes is crucial for maintaining insulin sensitivity.
Chromium can prevent lipid peroxidation induced by heat stress through its strong antioxidant activities, especially when it is added to the diet. It improves the action of insulin and nutrient metabolism through the activation of enzymes associated with such pathways.
In heat-stressed pigs, the addition of chromium (200 ppb) increased blood neutrophils by about 37%. In addition, chromium supplementation was observed to possibly have positive effects on both cell-mediated and humoral immunity in heat-stressed buffalo calves.
Over the past two decades, studies have shown with certainty that chromium supplementation affects many variables in chickens. Moreover, chromium is believed to increase insulin action in insulin-sensitive tissues (i.e. adipose and muscles), resulting in increased farm animal productivity through the improvement of feed intake, growth rate, carcass quality, reproductive parameters, and immune functions.
Betaine and heat stress
Trimethylglycine, also known as betaine because it was first isolated from the juice of sugar beets, accumulates in some animal, plant and microbial sources, and cells undergoing osmotic stress (Sizeland et al., 1993; Suzuki et al., 2003; Lever & Slow, 2010).
Betaine has three known functions in mammalian tissues:
- As an organic osmolyte, it helps to retain cell volume under osmotic stress and can accumulate in molar concentrations (Burg, 1995).
- It can protect against protein denaturation (Caldas et al., 1999).
- It can act as a methyl donor (Zeisel & Blusztajn, 1994).
It has never been formally quantified, but it is generally accepted that betaine is rapidly and nearly fully absorbed (Kettunen et al., 2001). When it is not catabolised, betaine can act as an organic osmoprotectant (Fernández et al., 1998; Huang et al., 2007) and is both actively and passively transported into the cell (Craig, 2004).
Dietary betaine supplementation can act as a carcass modifier by increasing carcass muscle and decreasing fat depths in pigs (Fernández-Figares et al., 2002; Huang et al., 2008). Additionally, betaine can enhance carnitine concentrations in liver and muscle tissues. As carnitine is required for fatty acid transport through mitochondrial membranes, betaine supplementation may improve fatty acid oxidation, thereby reducing carcass fat (Eklund et al., 2005). A decrease in fat thickness is particularly important in times of heat exposure as an increased subcutaneous fat depth can impair heat dissipation (Brown-Brandl et al., 2006; Gaughan et al., 2010).
Porcine muscle tissue accumulates betaine when it is supplemented in the diet, suggesting that these tissues would have increased water retention and perhaps a reduction in osmotic stress associated with heat dissipation methods (Matthews et al., 2001b).
Furthermore, the capacity of betaine to utilise osmotic gradients rather than ion pumps in intestinal tissues, can decrease the energy required for Na+/K+ pumps by 60% (Moeckel et al., 2002), potentially allowing whole-body energy savings of approximately 8% (Cronje, 2005) and reducing metabolic heat production. In pigs that have the potential to use this additional energy, such as those restrictively fed and/or in porcine somatrotopin-treated boars, the additional energy can be used for lean tissue deposition (Suster et al., 2004).
In one study, total heat production was lower in pigs fed an energy-limiting diet supplemented with betaine (1,25g/kg feed) and housed in thermoneutral (20°C, 65% relative humidity) conditions. This can be attributed to either an improvement in efficiency or a decrease in maintenance energy requirements (Schrama et al., 2003).
Mashilo Phosa is the Swine team lead at Chemuniqué











Thanks for the information is vaulable